The halogen atom takes an electron from metal atom. Halogens form ionic bonds with other metal _atoms _when they react. Halogens form covalent bonds with other non-metal atoms when they react. As you move down the group, the amount of electron shielding increases, meaning that the electron is less attracted to the nucleus. This is because group 7 elements react by gaining an electron. Relative atomic mass increases down the group. This is because the atomic radii increase down the group, increasing the amount of intermolecular forces holding each molecule together. Melting/boiling points increase down the group. For this reason, fluorine is the most reactive halogen and astatine is the least reactive of the halogens. How relative atomic mass increase down group 7 and why. How melting and boiling points increase down group 7 and why. How group 7 elements react with metals and nonmetals. Group 1Reactions with Metals and Non Metals 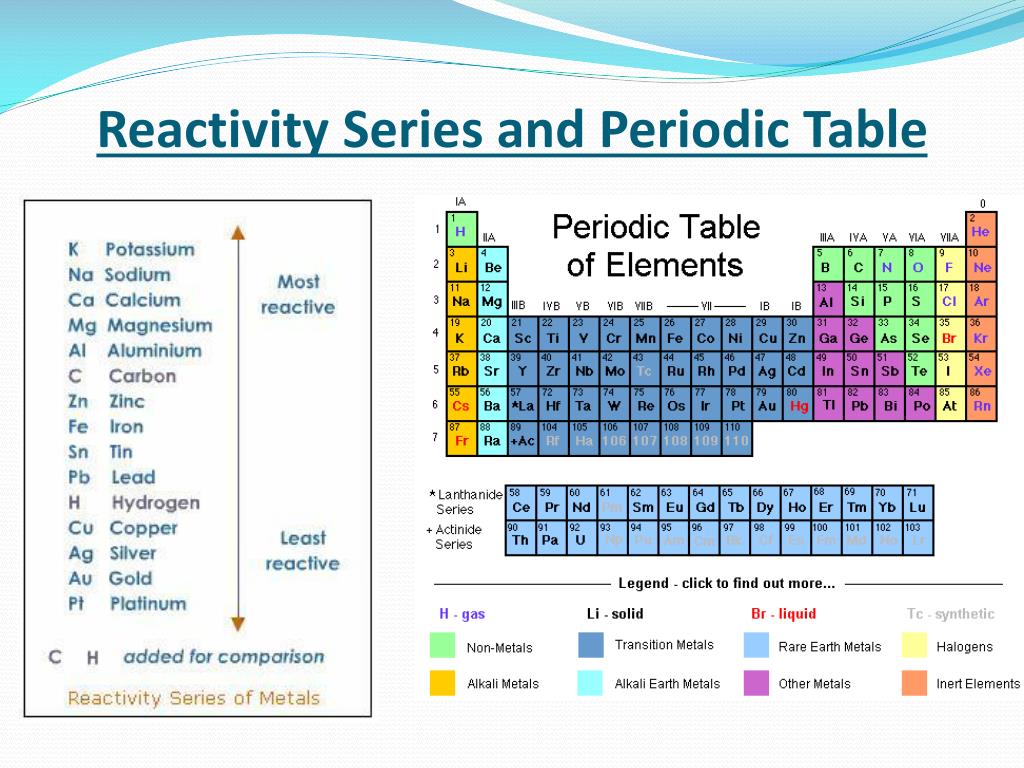
Iodine is a poisonous grey solid or a purple gas.Bromine is a poisonous brown liquid or orange gas.They are coloured vapours at room temperature: They are diatomic (travel in pairs, i.e Br2) Group 7 elements are known as the halogens. Combined Science: Rate of Chemical Change. 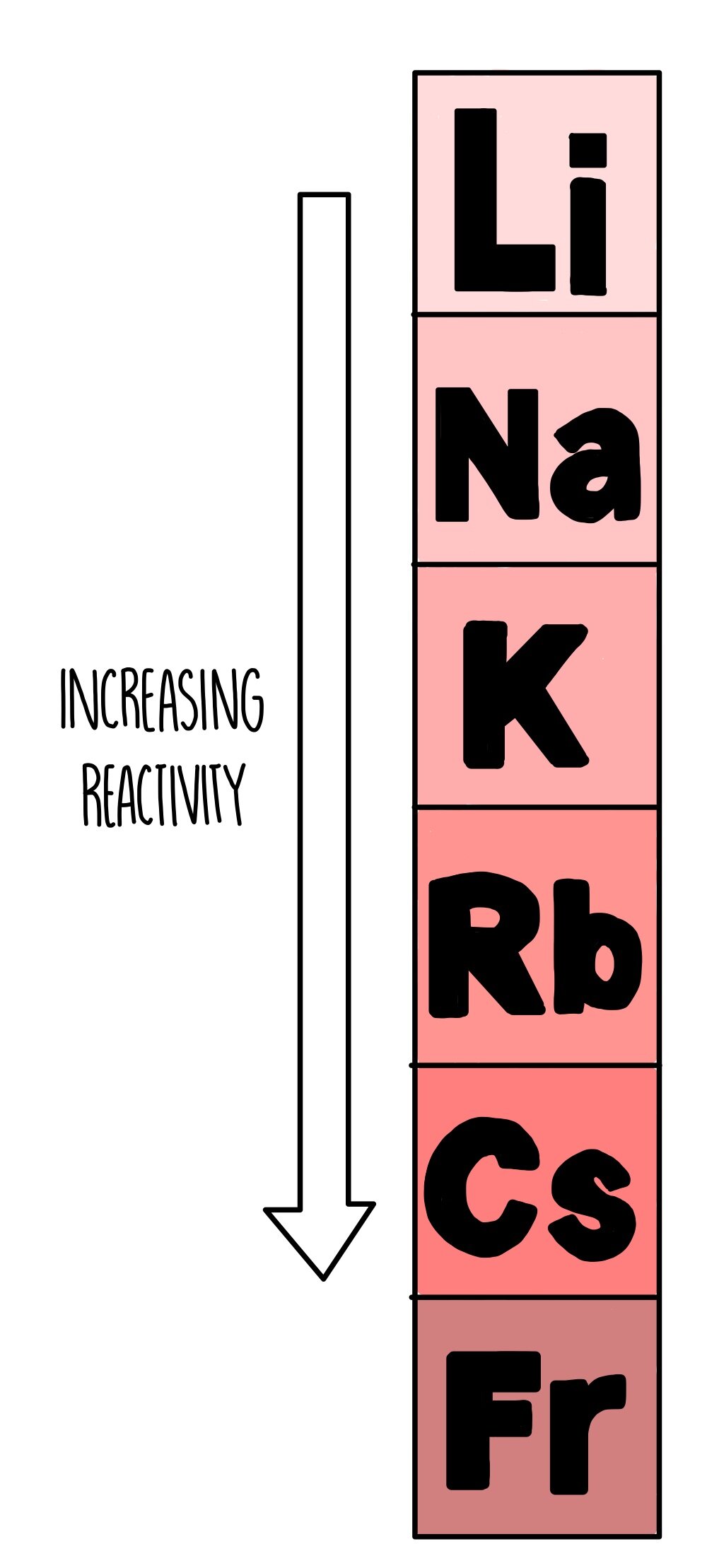
Combined Science: Atmospheric Chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
